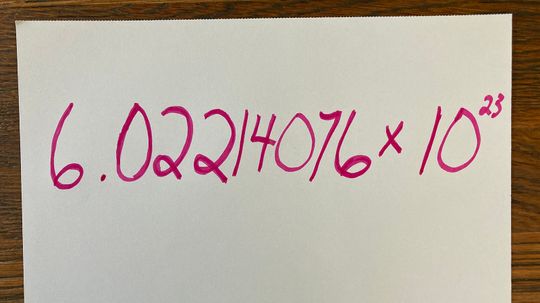Chemistry
Chemistry is the science of matter and the changes it undergoes during chemical reactions. In this section, learn about everyday chemistry, from chlorine beach to helium, and even why chocolate turns gray.

It's Elementary: The Periodic Table Quiz

Alkali Metals: Elements in the First Column of the Periodic Table

Cadmium: The Highly Toxic Metal That Powers the World
Strong Bases: Properties, Applications and Examples

Comparing Strong Acids and Weak Acids

10 Things You Should Never Mix With Alcohol

Delta-8 vs. Delta-9: Comparing Types of THC

What Color Is the Hottest Flame?

Why Do Bubbles Pop?
Learn More
"Delta 8" has become a bit of a buzzword in the cannabis industry and the community health sphere. But what exactly is delta-8, and how is it different from "regular" cannabis?
By Sascha Bos
In chemistry, the classification of substances into acids and bases is fundamental.
By Marie Look
In the world of chemistry, understanding the difference between strong acids and weak acids is fundamental for both students and professionals alike. Strong acids are known for their ability to completely dissociate in water, making them a pivotal topic in chemical reactions and laboratory experiments.
Advertisement
Flame colors span a spectrum that tells a tale as old as fire itself. Many people wonder what color is the hottest flame; more than a testament to the natural fascination with fire's beauty, this question underscores a fundamental principle in the science of thermodynamics and combustion.
All bubbles pop — that's a fact of life. But what's the science behind the short life and inevitable pop of a bubble?
It’s the ultimate cheat sheet for science class — and it’s right there hanging on the wall. What do you really know about the indispensable periodic table of elements?
Juice and soda mix well with alcohol, but a few things don't mix so well. Some may just produce embarrassing moments. Others could cost you your life.
By Beth Brindle
Advertisement
Why do newspapers turn yellow over time?
I have heard that carbon monoxide is extremely poisonous. Can you explain why?
Scenario: A helium balloon is up against the ceiling one day, and the next day it's on the floor. Does the balloon fall because the helium leaks out, or because the helium molecules slow down due to decreased pressure?
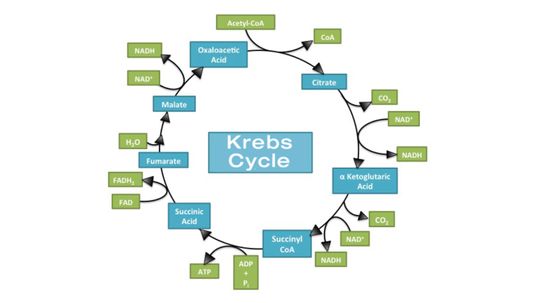
The main function of the Krebs cycle is to produce energy, stored and transported as ATP or GTP, to keep the human body up and running.
Advertisement
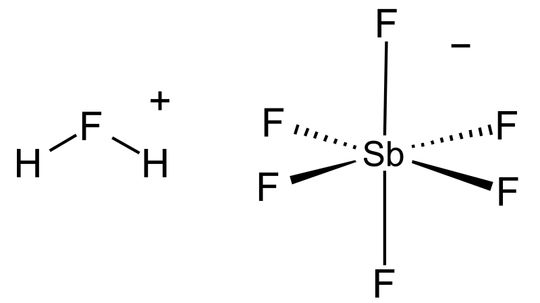
Superacids are those with an acidity greater than sulfuric acid. So which is the most super of superacids and what exactly is it used for?
Discovered in the early 1800s from a chunk of smuggled platinum ore, rhodium is the most valuable precious metal on the planet today, used mainly for keeping car emissions in check.
Cadmium is a natural metal and the leading component in rechargeable batteries and solar cells. It is also highly toxic and heavily regulated.
Once considered a semiprecious metal alongside gold and silver, aluminum pretty much languished in obscurity until the 19th century. How did the metal become so ubiquitous?
Advertisement
First there was Volvo. Then came IKEA. Well get ready for the next major Swedish export: snus, a smokeless tobacco product, similar to dip or chew.
Marijuana isn't just a recreational drug for hippies and philosophy majors -- its psychoactive history ranges from Egyptian mummies to modern U.S. politics. What's the big deal about this leafy, green plant?
By Kevin Bonsor & Nicholas Gerbis
You know how chocolate sometimes turns white? Why does that happen and is it still OK to eat?
Have you seen investigators on crime shows who spray some stuff on a "clean" carpet and suddenly -- blood stains! Well, of all the fictional technology on TV, it turns out this stuff is real! Find out how luminol reveals the blood.
By Tom Harris
Advertisement
More than 2.3 billion people across the globe drink alcohol, but most don't consider it a drug. But if you've ever seen someone who's had too much, you know alcohol has profound effects on the mind and body.
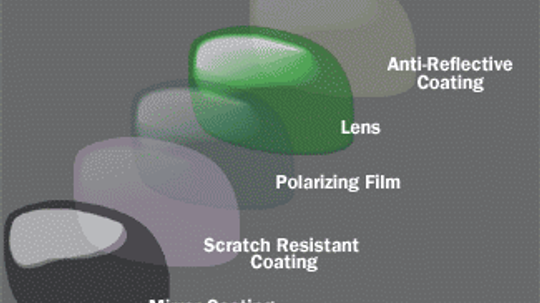
I recently bought a pair of mirrored sunglasses and they are already scratched. Isn't there a way to make them scratch-resistant?
Imagine spending your days racked with pain or losing pound upon pound because nausea leaves you unable to eat. Now imagine that someone offers you a wonder drug to cure all your ills. The problem? It's illegal.
That's one seriously big number, and technically Amedeo Avogadro didn't even come up with it. So how did the Italian chemist make such an indelible (numerical) mark on the wonderful world of chemistry?
Advertisement
Can you pass the acid test? That electric Kool-Aid changed the fabric of 1960's American counterculture. So, what's it's like to trip on LSD?
Smoking or chewing tobacco makes many people feel good, even mildly euphoric. It's the nicotine that produces the buzz. Find out how nicotine affects the human body and what makes it so addictive.