Key Takeaways
- Electronic cigarettes work by heating a nicotine liquid and converting it into a vapor, avoiding the combustion process of traditional cigarettes.
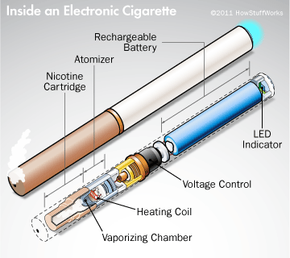
- They consist of three main parts: a rechargeable lithium battery, a vaporization chamber and a cartridge containing nicotine liquid.
- Despite their popularity and perceived benefits over traditional smoking, health experts express concerns about their safety and the promotion of e-cigarettes as a healthier alternative.
You're at your favorite restaurant, enjoying a meal. A diner at the next table is puffing on a cigarette, letting out a cloud of smoke. Because smoking isn't allowed in the restaurant, you're thinking about asking the smoker to put the cigarette out. But before you protest, consider this: Your neighbor may not be smoking at all.
Electronic cigarettes, also known as smokeless cigarettes, e-cigarettes, or e-cigs, are an alternative method of consuming nicotine, the addictive chemical found in tobacco. Manufacturers often design e-cigarettes to look like regular cigarettes, but they contain no tobacco and don't require a match -- or any flame at all.
Advertisement
An e-cigarette is a battery-powered device that converts liquid nicotine into a mist, or vapor, that the user inhales. There's no fire, no ash and no smoky smell. E-cigarettes do not contain all of the harmful chemicals associated with smoking tobacco cigarettes, such as carbon monoxide and tar.
Manufacturers and satisfied customers say the e-cigarette is a healthier alternative to tobacco cigarettes, which cause millions of deaths every year. Some users say e-cigs have helped reduce their "smoker's cough," sharpened their senses of taste and smell, and even improved their sleep.
The electronic cigarette was invented by Chinese pharmacist Hon Lik, who patented the device in 2003 and introduced it to the Chinese market the following year. Numerous companies are now selling e-cigarettes to customers around the world. But as e-cigarette smoking -- or "vaping" as it's sometimes called -- has grown in popularity, some have concerns about its safety, including the possibility that the vapor created by the devices contains dangerous chemicals.
Is the electronic cigarette a cleaner, healthier choice for smokers? Or is it a dangerous device with hidden risks? Both viewpoints have their merits, but on the next page we'll start with the basics: how the product works, and why it's popular.
Advertisement