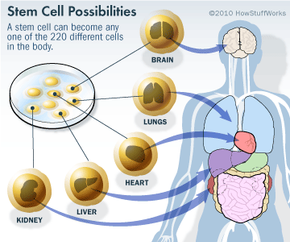
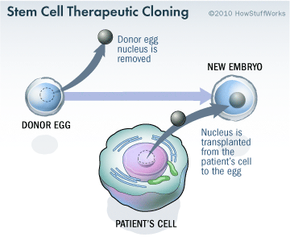
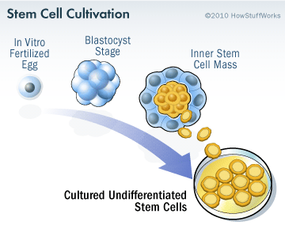
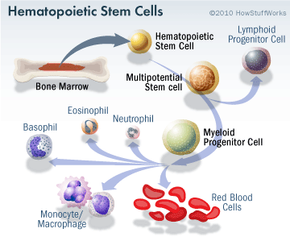
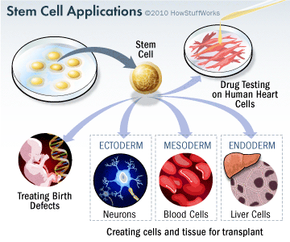
Stem cell research has become one of the biggest issues dividing the scientific and religious communities around the world. At the core of the issue is one central question: When does life begin? At this time, to get stem cells that are reliable, scientists either have to use an embryo that has already been conceived or else clone an embryo using a cell from a patient's body and a donated egg. Either way, to harvest an embryo's stem cells, scientists must destroy it. Although that embryo may only contain four or five cells, some religious leaders say that destroying it is the equivalent of taking a human life. Inevitably, this issue entered the political arena.
In 1996, Congress passed a rider to the federal appropriations bill called the Dickey-Wicker amendment. Representatives Jay Dickey and Roger Wicker proposed banning the use of federal monies for any research in which a human embryo is created or destroyed. Federal monies are a primary source of funding for stem cell research. The amendment has been renewed every year since that time.
In 2001, President George W. Bush further restricted federal stem cell research. In an executive order, Bush stated that federal funds could only be used for research on human embryonic stem cell lines that had already been established (only 22 cell lines). This prevented researchers from creating more embryonic stem cell lines for research.
In 2009, President Barack Obama issued an executive order to expand embryonic stem cell research. Obama's administration allowed federal funding of embryonic stem cell research if the following conditions applied:
- The cell line was one of the 22 in existence during the Bush administration or was created from embryos that had been discarded after in vitro fertilization procedures.
- The donors of the embryos were not paid in any way.
- The donors clearly knew that the embryos would be used for research purposes prior to giving consent.
According to the administration, the new policy did not violate the Dickey-Wicker amendment because the money did not finance the creation of new embryos (they had already been created by private means) and did not finance the destruction of them.
In 2009, two researchers from Boston, Dr. James Sherley of the Boston Biomedical Research Institute and Dr. Theresa Deisher of the Ava Maria Biotechnology Company, and other agencies filed a lawsuit against the government. Initially, the lawsuit was dismissed because the judge ruled that the plaintiffs had no legal standing (i.e. they were not affected materially by the new rules). However, a court of appeals overturned the initial ruling. The two scientists remained plaintiffs. The scientists claimed that, because they used adult stem cells exclusively in their research, the new rules would increase competition for federal research dollars, thereby affecting their ability to obtain funding. Federal Judge Royce Lamberth upheld the appeals court ruling. He placed an injunction preventing the new rules from going into place. He claimed that the rules violated the Dickey-Wicker amendment because embryos must be destroyed in the process of creating embryonic stem cell lines.
In September 2010, The New York Times reported that the U.S. Court of Appeals ruled that federal funding of embryonic stem cell research could continue under the new rules while the court considers Judge Lamberth's ruling [source: New York Times]. This ruling allows researchers to continue feeding embryonic stem cell cultures, experimenting with mice, and other research activities until this court rules, the U.S. Supreme Court weighs in, or Congress passes legislation that clarifies the issues. In the meantime, stem cell research and the careers of stem cell researchers hang on a legal roller coaster. Although stem cells have great potential for treating diseases, much work on the science, ethical and legal fronts remains.
For more on stem cells, investigate the links on the following page.