Key Takeaways
- The AbioCor artificial could potentially the double life expectancies of patients with end-stage heart failure.
- Candidates for the AbioCor artificial heart must have end-stage heart failure, a life expectancy of less than 30 days, and no other viable treatment options.
- The first recipient of the AbioCor artificial heart was Robert Tools, who, despite battling an infection and needing a ventilator post-surgery, had his mechanical heart continue to function without problems.
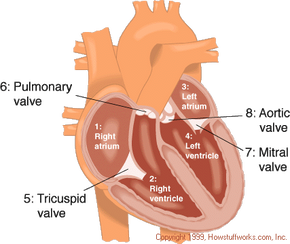
Your heart is the engine inside your body that keeps everything running. Basically, the heart is a muscular pump that maintains oxygen and blood circulation through your lungs and body. In a day, your heart pumps about 2,000 gallons of blood. Like any engine, if the heart is not well taken care of it can break down and pump less efficiently, a condition called heart failure.
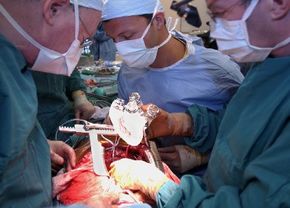
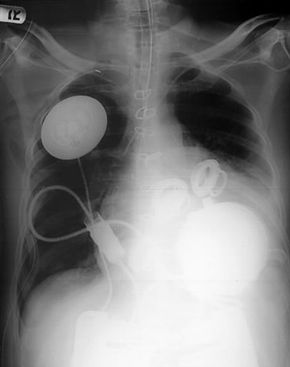
For a long time, the only option for many severe heart failure patients was a heart transplant. However, there are only slightly more than 2,000 heart transplants performed in the United States annually, meaning that tens of thousands of people die waiting for a donor heart. On July 2, 2001, heart failure patients were given new hope as surgeons at Jewish Hospital in Louisville, Kentucky, performed the first artificial heart transplant in nearly two decades. The AbioCor Implantable Replacement Heart was the first completely self-contained artificial heart and is expected to at least double the life expectancy of heart patients.
Advertisement
In this article, you will get an in-depth look at how this artificial heart works, how it's implanted into a patient's chest and who might be a candidate for receiving one of these mechanical hearts. We will also compare the AbioCor heart to the artificial hearts that have failed in the past.
Advertisement