There's a lot of chemistry happening inside a chloroplast, but the result of the chemistry is the conversion of sunlight into stored energy — basically the creation of a battery.
So, let's follow the energy:
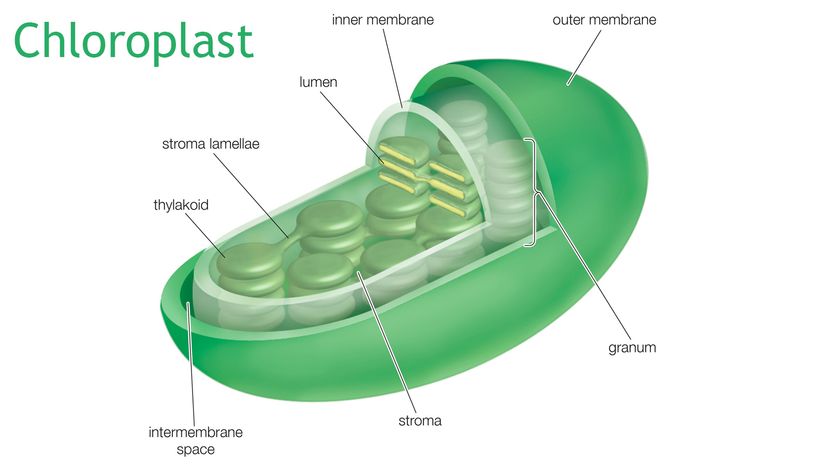
The sun shines on a leaf. That solar energy excites electrons inside water molecules in the leaf, and because excited electrons bounce around a lot, the hydrogen and oxygen atoms in the water molecules break apart, launching these excited electrons into the first stage of photosynthesis — a conglomeration of enzymes, proteins and pigments called photosystem II, which breaks down water, producing hydrogen ions (protons that will be used in the battery and oxygen gas that will float off into the air as plant garbage).
These energized electrons get passed on to some other membrane-bound proteins that use that energy to power ion pumps that escort the hydrogen ions from the space between the membranes into the thylakoid space, which is where all the light-dependent reactions of photosynthesis occur. Photosystems and electron pumps cover the surfaces of the thylakoid membranes, pumping the hydrogen ions from the stroma (the fluid space between the thylakoid and the inner membrane) into the stacks and stacks of thylakoid pouches — and these ions really want to get out of these thylakoids, which is what creates the electrochemical gradient. In this way light energy — that stuff that shines on your face when you go outside — is converted into a sort of battery, like the ones that run your wireless earbuds.
At this point, photosystem I takes over, which arranges for temporary storage of the energy generated by the battery. Now that the electron has been allowed to move along the gradient, it's much more relaxed, so it absorbs some light to re-energize it, and passes that energy along to a special enzyme that uses it, the electron itself, and a spare proton to make NADPH, which is an energy-carrying molecule that provides short-term storage for chemical energy that will later be used to make glucose.
At this point, the light energy is now in two places: It's stored in the NADPH and as the electro-chemical gradient of the difference in hydrogen ion concentration inside the thylakoid compared to just outside it in the stroma.
"But the high hydrogen ion gradient inside the thylakoid wants to degrade — it needs to degrade," says Jackson. "Gradients represent 'organization' — essentially the opposite of entropy. And thermodynamics tells us that entropy will always try to increase, which means that a gradient must break down. So, the hydrogen ions inside each thylakoid really want to escape in order to even out the concentrations on either side of that inner membrane. But charged particles can't pass through a phospholipid bilayer just anywhere — they need some kind of channel to go through, just like electrons need a wire to make it from one side of the battery to the other."
So, just like you can put an electric motor on that wire, and make electrons drive a car, the channel the hydrogen ions pass through is a motor. These protons flow through the channel provided for them, like water flowing through a hydroelectric dam down an elevational gradient, and that motion makes enough energy to create a reaction that creates ATP, which is another short-term storage form of energy.
Now the original light energy has been converted into short-term storage chemical energy in the form of both NADPH and ATP, which will be useful later in the dark reactions (also known as the Calvin Cycle or the carbon-fixation cycle) within the chloroplast, all of which go down in the stroma because this fluid contains an enzyme that can convert NADPH, ATP and carbon dioxide into sugars that either feed the plant, assist in respiration, or are used to produce cellulose.
"Complex organic molecules like cellulose, which is made of glucose, take a lot of energy to make, and that all came from the sun," says Jackson. "Following the energy, it starts as light wave energy, then excited electron energy, then electrochemical gradient energy, then chemical energy in the form of NADPH and ATP. The oxygen gas is breathed off, and the NADPH and ATP aren't used to do other stuff within the cell — instead, both are passed to the carbon-fixation cycle, where other enzymes break them down, extract that energy, and use it to build glucose and other organic molecules."
And all this, thanks to a little organelle called a chloroplast.