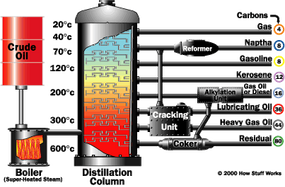
Chemical Processing
You can change one fraction into another by one of three methods:
- breaking large hydrocarbons into smaller pieces (cracking)
- combining smaller pieces to make larger ones (unification)
- rearranging various pieces to make desired hydrocarbons (alteration)
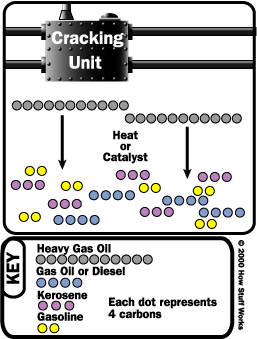
Cracking
Cracking takes large hydrocarbons and breaks them into smaller ones.
Advertisement
There are several types of cracking:
- Thermal - you heat large hydrocarbons at high temperatures (sometimes high pressures as well) until they break apart. steam - high temperature steam (1500 degrees Fahrenheit / 816 degrees Celsius) is used to break ethane, butane and naptha into ethylene and benzene, which are used to manufacture chemicals. visbreaking - residual from the distillation tower is heated (900 degrees Fahrenheit / 482 degrees Celsius), cooled with gas oil and rapidly burned (flashed) in a distillation tower. This process reduces the viscosity of heavy weight oils and produces tar. coking - residual from the distillation tower is heated to temperatures above 900 degrees Fahrenheit / 482 degrees Celsius until it cracks into heavy oil, gasoline and naphtha. When the process is done, a heavy, almost pure carbon residue is left (coke); the coke is cleaned from the cokers and sold. Photo courtesy Phillips Petroleum Company Catalysts used in catalytic cracking or reforming
- Catalytic - uses a catalyst to speed up the cracking reaction. Catalysts include zeolite, aluminum hydrosilicate, bauxite and silica-alumina. fluid catalytic cracking - a hot, fluid catalyst (1000 degrees Fahrenheit / 538 degrees Celsius) cracks heavy gas oil into diesel oils and gasoline. hydrocracking - similar to fluid catalytic cracking, but uses a different catalyst, lower temperatures, higher pressure, and hydrogen gas. It takes heavy oil and cracks it into gasoline and kerosene (jet fuel).
After various hydrocarbons are cracked into smaller hydrocarbons, the products go through another fractional distillation column to separate them.
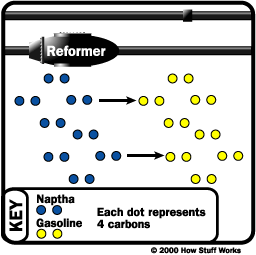
Unification
Sometimes, you need to combine smaller hydrocarbons to make larger ones -- this process is called unification. The major unification process is called catalytic reforming and uses a catalyst (platinum, platinum-rhenium mix) to combine low weight naphtha into aromatics, which are used in making chemicals and in blending gasoline. A significant by-product of this reaction is hydrogen gas, which is then either used for hydrocracking or sold.
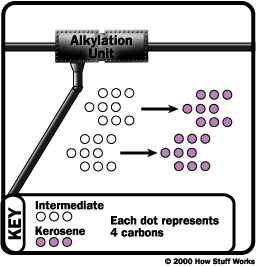
Alteration
Sometimes, the structures of molecules in one fraction are rearranged to produce another. Commonly, this is done using a process called alkylation. In alkylation, low molecular weight compounds, such as propylene and butylene, are mixed in the presence of a catalyst such as hydrofluoric acid or sulfuric acid (a by-product from removing impurities from many oil products). The products of alkylation are high octane hydrocarbons, which are used in gasoline blends to reduce knocking (see "What does octane mean?" for details).
Now that we have seen how various fractions are changed, we will discuss the how the fractions are treated and blended to make commercial products.