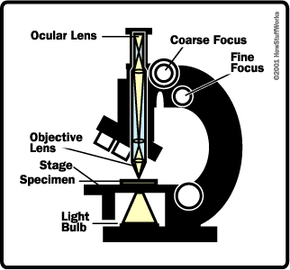
A light microscope works very much like a refracting telescope, but with some minor differences. Let's briefly review how a telescope works.
A telescope must gather large amounts of light from a dim, distant object; therefore, it needs a large objective lens to gather as much light as possible and bring it to a bright focus. Because the objective lens is large, it brings the image of the object to a focus at some distance away, which is why telescopes are much longer than microscopes. The eyepiece of the telescope then magnifies that image as it brings it to your eye.
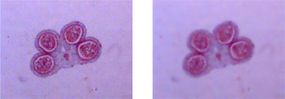
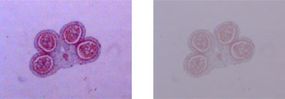
In contrast to a telescope, a microscope must gather light from a tiny area of a thin, well-illuminated specimen that is close-by. So the microscope does not need a large objective lens. Instead, the objective lens of a microscope is small and spherical, which means that it has a much shorter focal length on either side. It brings the image of the object into focus at a short distance within the microscope's tube. The image is then magnified by a second lens, called an ocular lens or eyepiece, as it is brought to your eye.
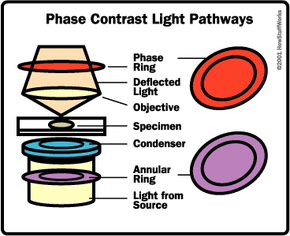
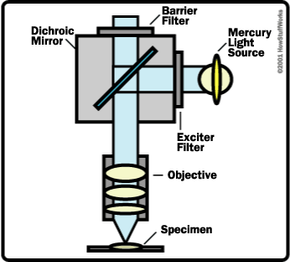
The other major difference between a telescope and a microscope is that a microscope has a light source and a condenser. The condenser is a lens system that focuses the light from the source onto a tiny, bright spot of the specimen, which is the same area that the objective lens examines.
Also unlike a telescope, which has a fixed objective lens and interchangeable eyepieces, microscopes typically have interchangeable objective lenses and fixed eyepieces. By changing the objective lenses (going from relatively flat, low-magnification objectives to rounder, high-magnification objectives), a microscope can bring increasingly smaller areas into view -- light gathering is not the primary task of a microscope's objective lens, as it is a telescope's.
We'll take a detailed look at the parts of a microscope later in the article.