Key Takeaways
- Boyle's Law describes the relationship between the pressure and volume of a gas.
- It states that at a constant temperature, the pressure of a gas is inversely proportional to its volume.
- This law is fundamental in understanding gas behavior and is widely applied in various fields such as chemistry, physics and engineering.
At some point in your life, you probably have taken — or will eventually take — a physics class. This is for your own good, although it's possible you might not feel the truth of that at the time. One of the reasons physics class is valuable has to do with putting a name and a picture to stuff you already know.
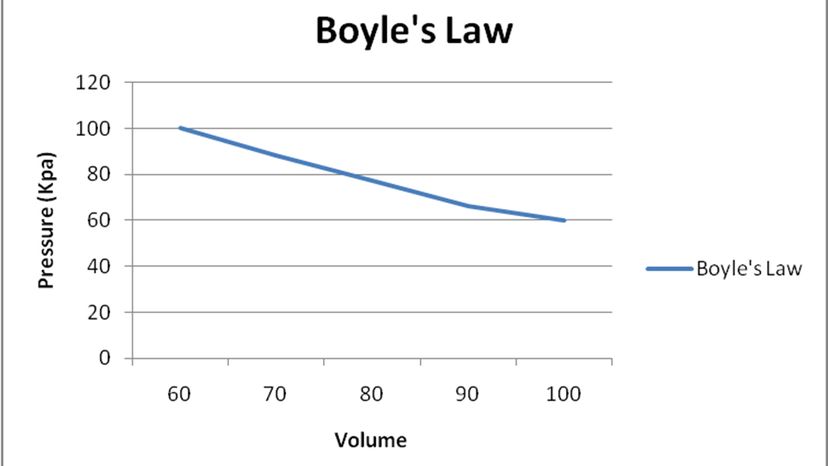
Take Boyle's Law, one of the gas laws that govern the way temperature, volume and pressure affect gases. Boyle's Law, named for Robert Boyle, the 17th century scientist who first published about it, is a description of the relationship between the pressure and volume of gas in a container (given the temperature remains constant). Your eyes might have just glazed over, but you already know all about Boyle's Law, and this is why you need physics class to put a name to what you already found out when you popped your first balloon. Sometimes science is just a way of making connections between what you know and we know, collectively.
Advertisement
Boyle's Law says there's an inverse relationship between the volume inside a container filled with gas and the pressure that gas is under. For instance, say you blew up a balloon only about half full of gas — in this case, the gas is the mixture of water vapor and carbon dioxide that comes out of our lungs when we exhale.
In the half-full balloon there's plenty of space for all those gas atoms to bump around without crowding any of them very much — the balloon itself might be pretty floppy. However, when you pinch all the air into one end of the balloon, the part of the balloon that has gas in it becomes taut. The number of gas molecules in the balloon hasn't changed — just the size of the container. If you then kept decreasing the size of the container, the pressure inside the balloon would increase to the point that the balloon would eventually pop.
Boyle's Law says that as the volume in a container containing gas decreases, the pressure inside the container increases — and, conversely, as the volume increases, the pressure decreases. Boyle even came up with a mathematical formula to help us figure out the pressure or volume of a gas in a confined space: No matter how you change the pressure or volume in a container of gas, multiplying one by the other will yield a constant product.
Advertisement

