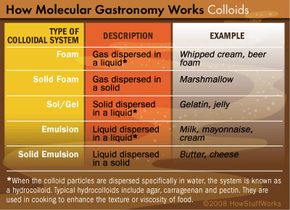
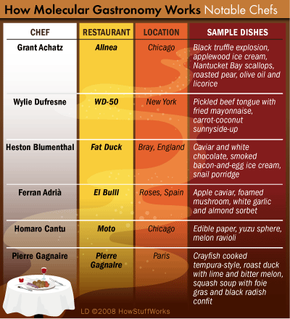
It's not the goal of molecular gastronomists to reduce cooking to a collection of dry calculations and lifeless formulas. Rather inventive cooks are trying to make their creations even tastier, with the help of a new technique or by tweaking an old favorite. Let's see how they might transform this traditional meal.
The main ingredient in caviar, the classic upscale hors d'oeuvre, is the eggs of certain fish species. With a little kitchen chemistry, you can enjoy a new kind of caviar — apple caviar — first developed by Ferran Adrià, who experimented with spherification.
Apple Caviar
Here's the basic recipe; you can find detailed instructions here.
Gather one-and-a-quarter pounds of golden apples, along with some alginate, baking soda, water and calcium chloride. Puree the golden apples, freeze for half an hour and then skim off the impurities and strain. Next, add the alginate to the apple juice while heating. Remove from heat and add the baking soda.
Now prepare a calcium chloride solution by dissolving calcium chloride in water. Finally, use a syringe to add your apple juice mixture to the calcium chloride solution one drop at a time. As you do, you should see beads, or "caviar," form. Cook for a minute in boiling water, strain and rinse in a cold-water bath.
Cointreau-infused Duck
For the main course, we're going to have duck à l'orange. The classic French recipe directs you to roast the bird in an oven for about two hours.
Roasting browns the meat and adds flavor through a series of chemical changes known as Maillard reactions. These reactions cause sugars and amino acids in the meat to cross-link. This, in turn, creates the compounds responsible for the pleasing color and flavor.
Unfortunately, cooking meat at high temperatures also has some negative effects. Most notably, the muscle fibers contract and shorten, forcing out water and making the meat tougher.
A molecular gastronomist overcomes this by taking advantage of microwave technology. When prepared in a microwave, meat warms to 212 degrees F (100 degrees C) and remains at that temperature as long as it contains water.
Microwaving meat is faster and more efficient than roasting but doesn't produce the beneficial Maillard reactions. To get the best of both worlds, molecular gastronomists would brown the meat first in a skillet, inject Cointreau (an orange-flavored liqueur) into each piece with a syringe, then finish the cooking in the microwave.
Flash-frozen Vanilla Ice Cream
Homemade vanilla ice cream is last. The best ice cream has abundant air bubbles and small ice crystals, which makes the finished product light and smooth.
Traditionally, you would place your ingredients in an automatic ice cream maker to churn and freeze the mixture. Churning folds air into the material and breaks up ice crystals. But there's a limit to how cold an average machine can get. Most rely on your kitchen freezer, which reaches a temperature of 0 degrees F (-18 degrees C).
A molecular gastronomist uses a simpler technique: He or she pours liquid nitrogen directly into the ingredients, which will flash-freeze the mixture and create extra-small ice crystals that result in the smoothest ice cream possible.
If you're dying to make this classic dessert in a cutting-edge way, start with a basic recipe, like this one from the Food Network. After preparing the ice cream mixture, don your safety glasses and gloves and add liquid nitrogen while stirring with a wooden spoon. Stop when the ice cream reaches your desired thickness.