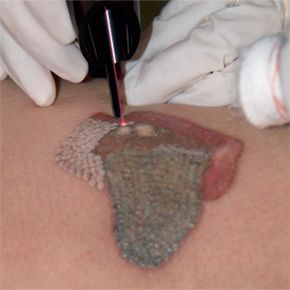
When Theodore Maiman fired the first laser pulse in 1960, some described the new technology as a solution in need of a problem. But scientists quickly discovered that lasers were not novelties and began developing practical applications for them. Today, physicians use lasers to repair damaged retinas, bleach birthmarks, remove regrettable tattoos and make delicate surgical cuts. The electronics industry incorporates lasers into a variety of components, including bar-code scanners, optical-storage systems and computer printers. And manufacturers harness the energy of lasers to drill holes in diamonds and cut materials ranging from titanium to plastic.
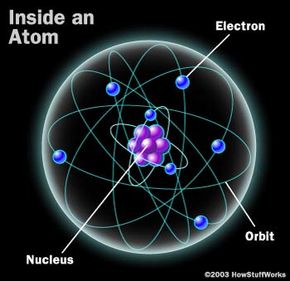
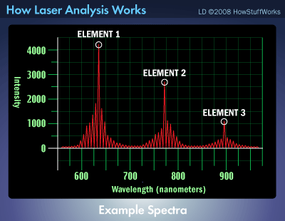
Lasers are particularly important to the field of analytical chemistry. Experts in analytical chemistry develop techniques to determine the chemical composition of substances. Some of these techniques measure physical properties, such as mass, refractive index or thermal conductivity. Other techniques rely on electric charges or current to help identify constituent parts of a substance. And still other methods measure the absorption, emission or scattering of electromagnetic radiation. This latter category is known as spectroscopy.
Advertisement
Laser-based spectroscopy is becoming an increasingly important analytical tool. Imagine a laser system mounted on a Mars-based rover. When it fires a laser pulse at Martian dirt, instrumentation on the rover detects the reflected light and determines the chemical makeup of the soil. Now imagine a soldier bearing a laser system mounted on his back. Using a hand-held probe that contains both laser and optics, the soldier analyzes a suspicious roadside package and determines it contains explosive material.
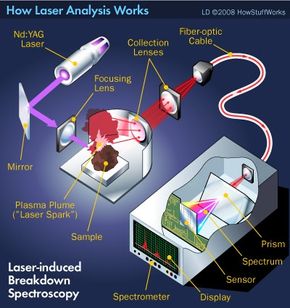
This kind of laser analysis seems like science fiction, but it's not. Scientists today have at their disposal many different types of laser-based analytical techniques. We'll explore some of those techniques in this article and examine one -- laser-induced breakdown spectroscopy, or LIBS -- in detail to illustrate the fundamentals of the technology. As we do, you'll learn how laser analysis is helping to advance everything from homeland security, forensics and medical diagnostics to health care, archeology and art history.
First, let's dive deeper into the basics of analytical chemistry to understand how laser technology fits into an arsenal of tools and techniques that can be used to determine the elemental or molecular building blocks of substances.