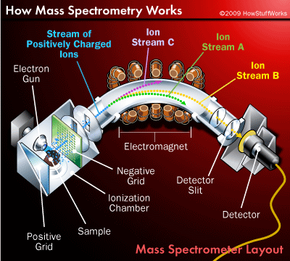
Deflection and Detection of Ions
Creating and accelerating ions are essentially preparatory steps to the real work of mass spectrometry -- mass analysis. The main job of the mass analyzer is to apply an external magnetic field to the ions exiting the ionization chamber. This external field interacts with the magnetic field generated by the fast-moving particles, causing the path of each particle to bend slightly. How much an ion's path curves depends on two factors: the mass of the ion and its charge. Lighter ions and ions with a greater charge are deflected more than heavier ions and ions with a smaller charge.
Chemists combine these two variables into a value called the mass-to-charge ratio, which is represented mathematically as m/z (or m/e). For example, if an ion has a mass of 18 units and a charge of 1+, its m/z value is 18. If an ion has a mass of 36 units and a 2+ charge, its m/z value is also 18. Most of the ions moving from the ionization chamber to the mass analyzer have lost a single electron, so they have a charge of 1+. That means the m/z value of most ions passing through a mass spectrometer is the same as the mass of the ion.
Advertisement
The net result is that each ion follows a path dependent on its mass, as shown on the right. Ion stream A has the lightest particles and is deflected the most. Ion stream C has the heaviest particles and is deflected the least. The mass of the particles in ion stream B falls somewhere in between. Notice that only one of the ion streams actually passes through the mass analyzer and reaches the detection unit at the back of the device. The other two streams hit the side of the spectrometer and are neutralized. To analyze all of the ions, chemists simply adjust the intensity of the magnetic field until each stream hits the detector.
A computer is used to analyze data collected at the detector and to display results in a graph known as a mass spectrum. On most graphs, the mass -- measured in atomic mass units (amu) -- is plotted on the x-axis. Relative intensity, which measures how many ions of a given mass are detected, is plotted on the y-axis.
Up next, we'll return to the major leagues and explore how mass spectrometry is used to expose steroid abuse in home-run sluggers.