Getting Ions Up to Speed: Understanding Mass Spectrometry
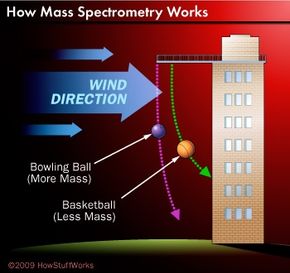
To understand the basic principles of mass spectrometry, consider a person standing at the top of a tower on a windy day. The person picks up various balls and drops them, one by one, from the tower. As each ball falls, wind deflects it along a curved path. The masses of the balls affect how they fall. A bowling ball, for example, is much heavier than a basketball and is therefore harder to move. As a result, a bowling ball follows a different path than a basketball.
In a mass spectrometer, the same thing is happening, except it's atoms and molecules that are being deflected, and it's electric or magnetic fields causing the deflection. It's also happening in a cabinet that can be as small as a microwave or as large as a chest freezer. The cabinet contains three basic parts: an ionization chamber, a mass analyzer and a detector. Here's how it all works.
Advertisement
To be deflected by electric or magnetic fields, atoms must first be ionized, or turned into charged particles. This is accomplished by knocking away one or more electrons, which results in a particle with a net positive charge. Spectrometrists sometimes bombard a sample with a beam of electrons to ionize the molecules. The incoming electrons act like billiard balls, knocking away electrons in the sample. They also use a technique known as electrospray ionization, which forces a sample through a charged needle to strip away electrons. Either way, this first step of mass spectrometry produces positive ions.
Next, the positive ions must move out of the ionization chamber. The force required to move them comes from an electric field supplied by two metal grids. One grid is positively charged and repels the ions; the other is negatively charged and attracts them. Because the repulsion and attraction act in the same direction, the ions move rapidly toward the negatively charged grid, which is perforated with many tiny holes. The ions pass through the holes at varying speeds. Lighter ions travel faster than heavier ones.
According to the laws of electromagnetism, a moving stream of electrically charged particles will generate a magnetic field. The ions in a mass spectrometer are no exception. They produce a magnetic field of their own, and it is this magnetic field that scientists take advantage of in the most important step of spectrometry. We'll get to that in the next section.