Electricity is the lifeblood of many aspects of our world. Without volts and amps, many of our technological innovations would cease to exist. Even our bodies wouldn't function without an electrical charge zipping through our cells. But what electricity gives, electricity can take away.
Although this form of energy is vital to so much of our lives, it's one of those things that are only good in the right amounts. Too much electricity can electrocute people. Likewise, it can kill our modern electronics and machines.
Advertisement
But thanks to Michael Faraday, the brilliant 19th-century scientist, and one of his namesake inventions, the Faraday cage, we humans have developed plenty of ways to control electricity and make it safer for our computers, cars and other inventions — and for us, too.
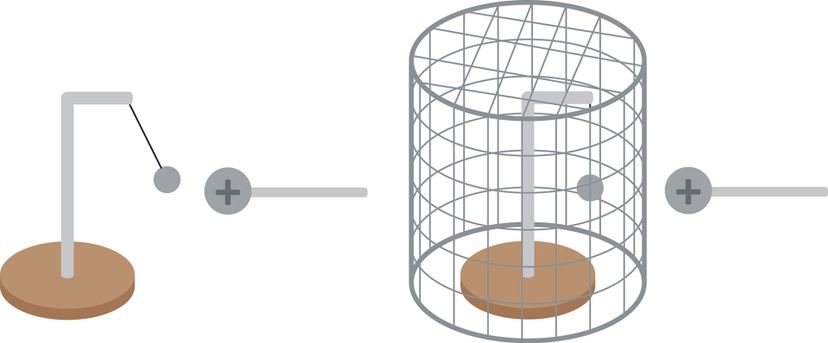
Faraday cages shield their contents from electromagnetic radiation. Basically, when an electromagnetic field hits something that can conduct electricity, the charges remain on the outside of the conductor rather than traveling inside. Faraday cages often look distinctly, well, cagelike. Some are as simple as chain-link fences or ice pails. Others use a fine metallic mesh.
Electromagnetic radiation is all around us. It's in visible and ultraviolet light, in the microwaves that cook our food and even in the FM and AM radio waves that pump music through our radios. But sometimes, this radiation is undesirable and downright disruptive. That's where Faraday cages come in.
As a Faraday cage distributes that charge or radiation around the cage's exterior, it cancels out electric charges or radiation within the cage's interior. In short, a Faraday cage is a hollow conductor, in which the charge remains on the external surface of the cage.
That basic function has plenty of fascinating uses in our electrically cluttered and technology-packed world. And although Faraday would eventually have his day, the backdrop for his invention actually has its roots in earlier times. So, where did the idea for these ultra-useful cages come from?
Advertisement