In its purest form, it's odorless, nearly colorless and tasteless. It's in your body, the food you eat and the beverages you drink. You use it to clean yourself, your clothes, your dishes, your car and everything else around you. You can travel on it or jump in it to cool off on hot summer days. Many of the products that you use every day contain it or were manufactured using it. All forms of life need it, and if they don't get enough of it, they die. Political disputes have centered around it. In some places, it's treasured and incredibly difficult to get. In others, it's incredibly easy to get and then squandered. What substance is more necessary to our existence than any other? Water.
At its most basic, water is a molecule with one oxygen atom and two hydrogen atoms, bonded together by shared electrons. It is a V-shaped polar molecule, which means that it's charged positively near the hydrogen atoms and negatively near the oxygen atom. Water molecules are naturally attracted and stick to each other because of this polarity, forming a hydrogen bond. This hydrogen bond is the reason behind many of water's special properties, such as the fact that it's denser in its liquid state than in its solid state (ice floats on water). We'll look closer at these special properties later.
Advertisement
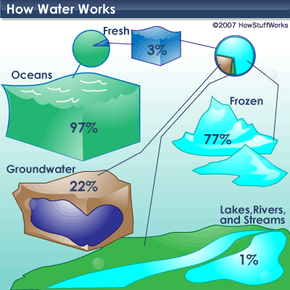
Water is the only substance that occurs naturally as a solid (ice), a liquid and a gas (water vapor). It covers about 70 percent of the Earth for a total of approximately 332.5 million cubic miles (1,386 million cubic kilometers) [source: U.S. Geological Survey]. If you're familiar with the lines "Water, water, everywhere, nor any drop to drink" from the poem "The Rime of the Ancient Mariner," you'll understand that most of this water -- 97 percent of it -- is undrinkable because it's saltwater (see illustration on next page). Only 3 percent of the world's water supply is freshwater, and 77 percent of that is frozen. Of the 23 percent that is not frozen, only a half a percent is available to supply every plant, animal and person on Earth with all the water they need to survive [source: National Geographic].
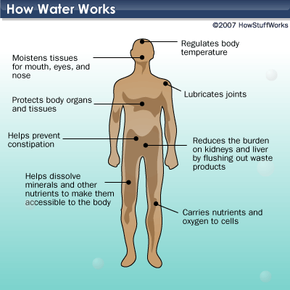
So water is pretty simple, right? Actually, there are a lot of things about it that scientists still don't fully understand. And the problem of making sure that enough clean, drinkable water is available to everyone and everything that needs it is anything but simple. In this article, we'll look at some of these problems. We'll also explore exactly what plants, animals and people do with water and learn more about what makes water so special.
Advertisement