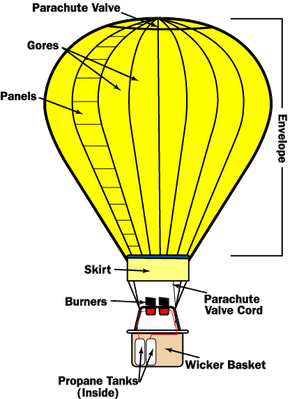
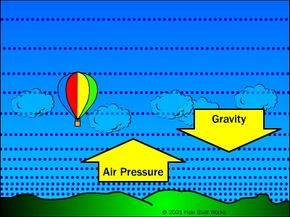
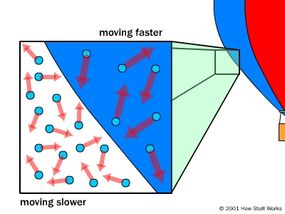
The basic idea behind hot air balloons has been around for a long time. Archemedes, one of the greatest mathematicians in Ancient Greece, figured out the principle of buoyancy more than 2,000 years ago, and may have conceived of flying machines lifted by the force. In the 13th century, the English scientist Roger Bacon and the German philosopher Albertus Magnus both proposed hypothetical flying machines based on the principle.
But nothing really got off the ground until the summer of 1783, when the Montgolfier brothers sent a sheep, a duck and a chicken on an eight-minute flight over France. The two brothers, Joseph and Etienne, worked for their family's prestigious paper company. As a side project, they began experimenting with paper vessels elevated by heated air.
Over the course of a couple years, they developed a hot air balloon very similar in design to the ones used today. But instead of using propane, they powered their model by burning straw, manure and other material in an attached fire pit.
The sheep, duck and chicken became the first balloon passengers on Sept. 19, 1783, in the Montgolfiers' first demonstration flight for King Louis XVI. They all survived the trip, giving the King some assurance that human beings could breath the atmosphere at the higher elevation. Two months later, the Marquis Francois d'Arlandes, a major in the infantry, and Pilatre de Rozier, a physics professor, became the first human beings to fly.
Other hot air balloon designs and ambitious flights followed, but by 1800, the hot air balloon had been largely overshadowed by gas balloons. One factor in this popularity decline was the death of Pilatre de Rozier in an attempted flight over the English Channel. The new balloon he built for the flight included a smaller hydrogen balloon in addition to the hot air balloon envelope. The fire ignited the hydrogen early in the flight, and the entire balloon burst into flames.
But the main reason hot air balloons fell out of fashion was that new gas balloon dirigible designs were superior in a number of ways — chiefly, they had longer flight times and could be steered.
Another popular balloon type was the smoke balloon. These balloons were lifted by a fire on the ground, and did not have any attached heat source. They simply shot up in the air, and then sank back to the ground.
Their main use was as an attraction at travelling fairs in the United States in the late 1800s and early 1900s. The balloonist would put on a parachute and attach himself to a canvas balloon. Then, several assistants would hold the balloon over a fire pit, getting the air hotter and hotter, and so increasing the upward force. When the force was great enough — and if the balloon hadn't caught on fire — the assistants would let go and the balloonist would be launched into the air. When the balloon reached its highest point, the balloonist would detach and parachute to the ground.
Since the 1960s, traditional hot air balloons have enjoyed a renaissance, due in part to a man named Ed Yost and his company, Raven Industries. Yost and his partners founded Raven Industries in 1956 to design and build hot air balloons for the United States Navy's Office of Naval Research (ONR). The ONR wanted the balloons for short-range transportation of small loads. Yost and his team took the basic concept of the Montgolfier brothers' balloon and expanded it, adding the propane burner system, new envelope material, a new inflation system and many important safety features.
They also came up with the modern, light-bulb-style envelope shape. Yost first designed large, spherical balloons. These balloons worked well, but had an odd inflation pattern: When the air was heated, the top of the balloon filled up, but the bottom stayed under-inflated. For efficiency, Yost just got rid of the extra fabric at the bottom, developing the familiar "natural" balloon shape we see today.
By the early 1960s, the ONR had lost interest in hot air balloons, so Yost began selling his balloons as sporting equipment. Other companies soon sprang up, as more and more people got involved in ballooning. Over the years, designers have continued to modify hot air balloons, adding new materials and safety features, as well as developing creative envelope shapes. Some manufacturers have also increased basket size and load capacity, building balloons that hold up to 20 passengers!
But the basic design is still Yost's modified version of the Montgolfier brothers' original concept. This remarkable technology has enthralled people all over the world. Balloon tours are a multi-million dollar business, and balloon races and other events continue to attract crowds of spectators and participants. It's even become fashionable (among billionaires) to build high-tech balloons for trips around the world. It really says a lot about hot air balloons that they are still so popular, even in the age of jet planes, helicopters and space shuttles.
For more information on hot air balloons and related topics, check out the links that follow.