To some, nuclear energy offers a clean energy alternative that frees us from the shackles of fossil fuel dependence. To others, it summons images of disaster: quake-ruptured Japanese power plants belching radioactive steam, the dead zone surrounding Chernobyl's concrete sarcophagus.
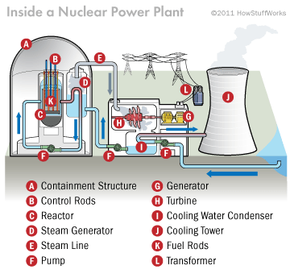
But what happens inside a nuclear power plant to bring such marvel and misery into being? Imagine following a volt of electricity back through the wall socket, all the way through miles of power lines to the nuclear reactor that generated it. You'd encounter the generator that produces the spark and the turbine that turns it. Next, you'd find the jet of steam that turns the turbine and finally the radioactive uranium bundle that heats water into steam. Welcome to the nuclear reactor core.
Advertisement
The water in the reactor also serves as a coolant for the radioactive material, preventing it from overheating and melting down. In March 2011, TV viewers around the world learned what happens when the cooling system suffers a catastrophic failure. Japanese citizens fled by the tens of thousands from the area surrounding the Fukushima-Daiichi nuclear facility after the most powerful earthquake on record and the ensuing tsunami inflicted serious damage on the plant and several of its reactor units. Among other events, water drained from the reactor core, which in turn made it impossible to control core temperatures. This resulted in overheating and a partial nuclear meltdown [source: NPR].
As of April 2018, there are about 450 nuclear power reactors in operation in 50 countries, and they provide about 11 percent of the world's electricity, according to the World Nuclear Association. In the U.S. alone, there are 99 reactors in 61 commercially operating nuclear power plants within 30 U.S. states, including Tennessee's Watts Bar Unit 2, a 1,150 megawatt-capacity reactor that began commercial operation in October 2016 [source: EIA].
Nuclear energy supplies 20 percent of U.S. electricity needs, less than the 31.7 percent that comes from natural gas and the 30.1 percent from coal, and only slightly more than the 17.1 percent provided by renewables such as hydropower, wind and solar [source: EIA]. But some countries depend on the atom more heavily. France, for example, gets 72 percent of its electricity from nuclear plants, and Sweden gets about 40 percent from them, according to a report from April 2018 [source: World-Nuclear.org].
In this article, we'll look at just how a nuclear reactor functions inside a power plant, as well as the atomic reaction that releases all that crucial heat.