Physical Science
Physical science is the study of the physical world around you. Learn about everything from electricity to magnetism in this section.
Brown Noise vs. White Noise: Which Is Best for Quality Sleep?

Can a sound wave kill you?

Can two cans and a string really be used to talk over a distance?

Delta-8 vs. Delta-9: Comparing Types of THC
Strong Bases: Properties, Applications and Examples

Comparing Strong Acids and Weak Acids

How Electricity Works

How Faraday Cages Work

How Gasoline Works

What do bugs have to do with forensic science?

5 Things You Didn't Know About Autopsies

Do a Person’s Fingerprints Change After Death?

How Alchemy Paved the Way for Chemistry

How did Nikola Tesla change the way we use energy?

Time May Not Exist, Say Some Physicists and Philosophers

Why Does Ice Stick to Your Fingers?
What if I forgot to remove a piercing before an MRI?

A Kid-friendly Introduction to Magnets and Magnetism

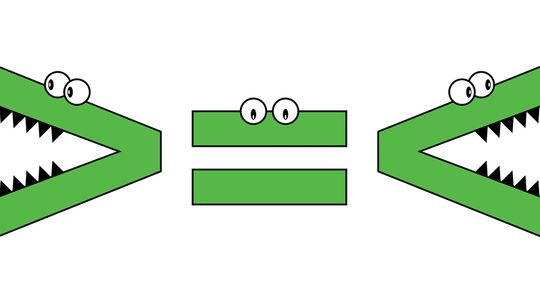
Deciphering 'Greater Than,' 'Less Than' and 'Equal To' Symbols

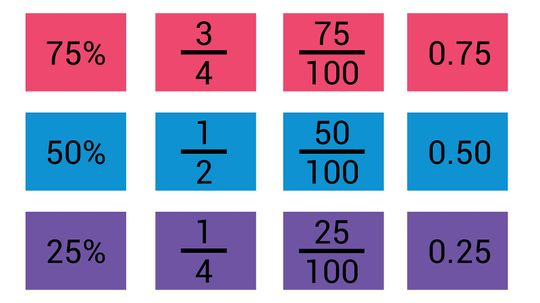
Getting a Handle on Fraction-to-Decimal Conversions

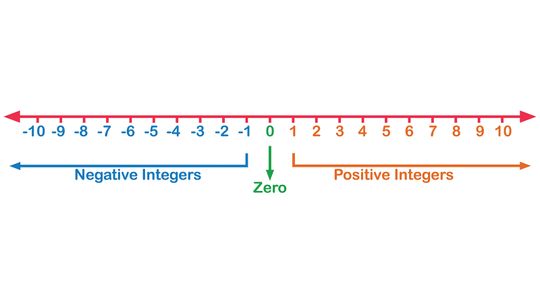
How to Make a Number Line for the Classroom

5 Hugely Fun Facts About Mass (Not Weight)

Antarctica's Spooky Cosmic Rays Might Shatter Physics As We Know It

Entropy: The Invisible Force That Brings Disorder to the Universe

The Demon Core: A Tale of Atomic Ambition and Tragic Fate

Half-Life Formula: Components and Applications

Could an 'X17 Particle' Hint at a Fifth Force in the Universe?

Why Are School Buses Yellow?

HowStuffWorks: How To Draw An Impossible Shape
What Are the Colors in the Visible Spectrum?
Learn More
Greater than, less than, equal to: These terms are mathematical expressions that allow the user to compare two numbers or equations. Once you've become familiar with these terms — and the symbols used to represent them — you'll be able to apply them to various math problems.
By Zach Taras
As you might recall from math class, fractions and decimals are two different ways of representing the same thing. A third option, percentages, is a close cousin of decimals. However, making use of this knowledge requires knowing how to convert one into the other.
By Zach Taras
"Delta 8" has become a bit of a buzzword in the cannabis industry and the community health sphere. But what exactly is delta-8, and how is it different from "regular" cannabis?
By Sascha Bos
Advertisement
A number line is a pictorial representation of real numbers. It is most commonly used in elementary math classes to help students compare numbers and perform arithmetic operations like addition, subtraction, division and multiplication.
By Mitch Ryan
Mean, median, mode and sometimes range, are all different methods for finding probability distribution in statistics. Range can be a helpful yardstick when calculating data values that are close together, but it can quickly become confusing if there is a wide gap between the smallest value and the largest number.
By Mitch Ryan
In chemistry, the classification of substances into acids and bases is fundamental.
By Marie Look
In the history of atomic research, few stories are as gripping or cautionary as that of the demon core, a plutonium sphere designed for one of history's most devastating weapons. This tale not only encapsulates the highest point of atomic ambition but also serves as a somber reminder of the human cost associated with such power.
Advertisement
In the world of chemistry, understanding the difference between strong acids and weak acids is fundamental for both students and professionals alike. Strong acids are known for their ability to completely dissociate in water, making them a pivotal topic in chemical reactions and laboratory experiments.
As a child, when trying to come up with the biggest number possible, you might have said "infinity plus one." While technically infinity is the largest number because you cannot run out of numbers, the biggest numbers that we know of are still difficult to count but a bit more quantifiable.
By Yara Simón
Flame colors span a spectrum that tells a tale as old as fire itself. Many people wonder what color is the hottest flame; more than a testament to the natural fascination with fire's beauty, this question underscores a fundamental principle in the science of thermodynamics and combustion.
Do you need to calculate the rate at which something changes over time? Whether it's the change in the x-value over the change in the y-value of a line on a graph, or the distance travelled by a car over the course of an hour-long drive, you'll need a rate of change formula.
By Sascha Bos
Advertisement
Physicists use the displacement formula to find an object's change in position. It sounds simple, but calculating displacement can quickly get complicated.
By Sascha Bos
In nuclear physics, the concept of half-life plays a crucial role in understanding the decay of radioactive substances. Scientists use the half-life formula in other disciplines to predict the rate of decay, as well as measure the age of ancient artifacts through carbon dating.
By Yara Simón
Frequency is a fundamental concept when you're talking about waves, whether that means electromagnetic waves like radio waves and visible light, or mechanical vibrations like sound waves.
By Marie Look
The wavelength formula is a fundamental concept in physics, particularly in the study of waves and electromagnetic radiation.
By Yara Simón
Advertisement
In math, few skills are as practical as knowing how to do long division. It's the art of breaking down complex problems into manageable steps, making it an essential tool for students and adults alike.
We get it: You need help with the parabola equation because those graphs won't draw themselves. Here's how to draw a parabola from an equation.
By Yara Simón
Trying to figure out whether your research problem would benefit from qualitative vs. quantitative data? Learn about the differences and uses of each.
By Yara Simón
Distinguishing between discrete vs. continuous data and situations that call for each data type is important in ensuring you get your desired results.
By Marie Look
Advertisement
The iconic "school bus yellow" was the invention of an educator named Frank Cyr. But if yellow is so good for visibility, why don't all fire trucks use it too?
By Dave Roos
All bubbles pop — that's a fact of life. But what's the science behind the short life and inevitable pop of a bubble?
The Standard Model of physics provides a framework for the subatomic world of all energies. Could a possible newfound carrier boson expand the definition of that framework?
By Mark Mancini
Whether you're a math whiz or not, there are some pretty cool number theories, beliefs and coincidences to appreciate. How down with digits are you?
By Alia Hoyt
Advertisement
You've probably had ice stick to your hand when you pull it out of the ice maker. But why is that?
By Alia Hoyt
It’s the ultimate cheat sheet for science class — and it’s right there hanging on the wall. What do you really know about the indispensable periodic table of elements?