Geology
Geology is the study of the composition and physical properties of rocks, minerals, gems and other related earth materials, including diamonds and crystals. Scientists gain an understanding of the Earth's history by studying its composition.

3 Types of Rocks Every Aspiring Geologist Should Know

What Are Geodes and Where Can You Find Them?

Is Africa Splitting in Two? Really? Here's the Scoop

15 Types of Gemstones to Add a Little Sparkle to Your Life

13 Brown Gemstones for Understated Elegance

10 Red Gemstones That Evoke Power and Bold Luxury

River-bottom Bones: The Strange World of Underwater Fossil Hunting

Velociraptor Alert: The Feathered Dinosaur Quiz

The Rockin' State Fossils Quiz
Learn More
From ancient amulets to modern engagement rings, different types of gemstones have always caught the human eye.
Looking to add earthy elegance to your jewelry box? Brown gemstones offer a unique and elegant alternative to more traditional colored gemstones.
Red gemstones bring a bold pop of color to any jewelry collection. From vibrant red hues to deep crimson tones, these stones have captivated wearers for centuries.
Advertisement
Yellow gemstones add a pop of sunshine to any jewelry collection. These stones, ranging from bright lemon to deep golden hues, can suit nearly every skin tone and style.
Pink gemstones blend science and sparkle like few other minerals. Whether you're drawn to the feminine hue for its metaphysical properties or just its stunning aesthetic, there's a pink stone for every style and budget.
Some gems dazzle with rainbow brilliance. Others whisper their power in deep, dark silence. Black gemstones aren't flashy, but they hold serious weight in style, symbolism and geology.
Purple gemstones aren't just for royalty, but their regal vibe is hard to ignore. From ancient talismans to modern crystal "therapy," purple stones have long held a prized place in our jewelry collections and cultural traditions.
Advertisement
Green is one of nature’s most captivating colors, and green gemstones bring that energy into jewelry with rich, varied tones. From deep emerald green to pale green and bluish green, these stones offer unmatched visual appeal.
From deep blue to light sky blue, blue gemstones offer a diverse range of hues that evoke serenity, power, and timeless beauty.
Rocks might look simple, but they tell an ancient story of Earth’s fiery depths, surface shifts and biological processes.
A subduction zone can shake things up — literally. These geological features are responsible for some of the most intense earthquakes, volcanic eruptions and tsunamis.
By Yara Simón
Advertisement
Imagine walking through a dusty, arid landscape when you stumble upon a seemingly ordinary rock. Its rough exterior might not catch the eye, but what lies within surely will. Geodes are one of nature's most spectacular treasures, offering a glimpse into the geological processes that shape our Earth.
Winding through the South Carolina low country, the Cooper River is a reed-lined haven for sportfish and shorebirds. The waterway originates in Berkeley County's Lake Moultrie. From there, it proceeds all the way down to Charleston, where it merges with the Ashley and the Wando to form that city's world-famous harbor. (Ever hear of Fort Sumter?)
By Mark Mancini
The notion of Africa splitting has the attention scientists and geologists worldwide, as the Great Rift Valley stretches and tears at the Earth's crust.
In the western Sahara Desert lies a natural wonder that has intrigued scientists and adventurers for centuries. Known as the Richat Structure - or, more commonly, the Eye of the Sahara - this massive geological formation resembles a giant eye.
By Marie Look
Advertisement
The Atacama Desert, situated in northern Chile, is not just any ordinary arid region. Spanning over 600 miles (965 km) along the Pacific Coast of South America, it is one of the most extreme landscapes on the planet. Thanks to certain oceanic conditions, there are areas that have received zero rainfall throughout recorded history, making the Atacama Desert the driest place on Earth.
By Marie Look
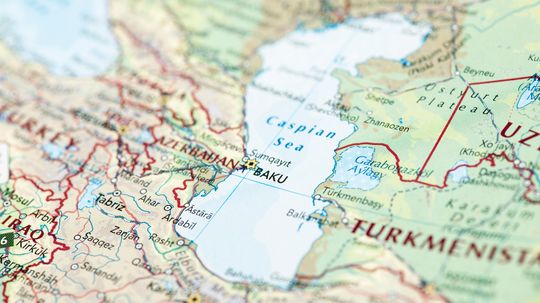
One of Earth's most interesting natural features, the Caspian Sea is the largest inland body of water on the planet, defying conventional classifications to be both a sea and a lake.
By Marie Look
Obsidian is more than just a beautiful stone. Discover its profound meaning and how it can serve as a powerful tool for healing and protection. Don’t miss out on the chance to learn about its transformative properties.
By HowStuffWorks
Discover the deep allure of smoky quartz meaning. Explore its grounding properties and metaphysical benefits. Unveil the mystique today.
By HowStuffWorks
Advertisement
Explore Aventurine meaning: a shimmering gem of luck and opportunity. Discover its healing properties and spiritual significance. Dive in now.
By HowStuffWorks
Uncover Pyrite meaning: a glittering symbol of wealth & protection. Explore its properties & spiritual significance. Delve into Pyrite's allure now.
By HowStuffWorks
Explore Chakra Crystals: Balancing energy centers for holistic wellness. Discover properties & benefits of crystal healing. Dive in now.
By HowStuffWorks
Explore Crystals for Love: Harness the energy of love-attracting stones. Discover meanings and benefits. Enhance your heart's resonance today.
By HowStuffWorks
Advertisement
Explore Crystal Grids: Amplify energy with sacred geometric arrangements. Learn about their uses and benefits. Elevate your intentions now.
By HowStuffWorks
Discover Crystals for Protection: Harness the power of protective stones. Explore their meanings and benefits. Enhance your energy shield today.
By HowStuffWorks